Experimental results
In this paper we report the results obtained with a process and apparatus not described here in detail and protected by patent in 90 countries, consisting of a system whose heat output is up to hundred times the electric energy input. As a consequence, the principle of the conservation of energy ensures that processes involving other energy forms are occurring in our apparatus.
The system on which we operate consists of Ni, in H atmosphere and in the presence of additives placed in a sealed container and heated by a current passing through a resistor. The maximum temperature value can be set to a wide range of values and an external meter allows us to measure the electric energy input. The container is in thermal contact with an external tank full of water and thermally insulated in order to minimize outside heat exchanges. As consequence of the energy production of the system, water boils and the water pipe is under pressure. The steam pressure cannot exceed a limit, whose value can be changed in the range 3-6 bar, because of the opening of a valve. When the valve opens, new water, whose amount is measured by a meter, enters from the supply. These data allow us to calculate the power produced by our system.
In stationary conditions the power output turns out to be much greater than the input (measured with an electric power meter). Some examples of the results obtained with this system (method A) in brief periods (~1-1,5 hours) are reported in lines 1-3 of the Table 1. The ratio between output and input power depends on changes occurring in the Ni-H system and on the time interval elapsed between the starting of the experiments and the measuring moments.
We have subsequently achieved a forced warm water movement through some radiators connected in series. In this case, the energy produced has been evaluated by measuring the power needed to obtain the same radiator temperature with a normal heating system (method B). In Table 1, lines 4 and 5, the results of these measurements are also reported. The patented apparatus is able of producing a constant and reliable amount of energy for a period of months.
A third method (method C) based on a closed circuit in which water is forced to circulate by means of a pump was used in order to measure the power generated: a section of the circuit contains the energy amplifier opportunely insulated in order to minimize thermal exchanges with outside.
Two thermocouples placed before and after the energy amplifier allow to detect continuously the water temperatures which are recorded on a computer. As a consequence the measured temperature difference allows to calculate the thermal energy transferred from the energy amplifier to the water. The electric input energy is measured by means an electric power meter.
In all cases the energy production is too high for any chemical process.

Table 1: Input and output energies, expressed in kWh, in some experiments.
(*) The anomaly in this experiment is due to contamination of the fuel.
In fact, assuming that each Ni atom in sample can realise, in optimal conditions, a typical chemical energy of some eV, the amount of energy emitted in the long lasting experiments would required at least 10ˆ28 atoms. That is something like a million of grams, a quantity enormously larger than the sample we have employed. For such a reason, we believe that form of energy involved is nuclear, and more specifically, due to fusion processes between protons and Nickel nuclei. They are exothermic with an energy release in the range 3-7,5 MeV, depending on the Nickel isotope involved.
It is remarkable that similar results have been obtained in the factory of EON in Bondeno (Ferrara, Italy) in a test performed with ENEL spa on June, 25th 2009 and in another sery of tests made in Bedford, New Hampshire (USA) in a lab of LTI with the presence of the DOE (November 19 2009) and of the DOD (November 20 2009).
The proton capture process performed by a Nickel nucleus produces a Copper nucleus according to the scheme
![]() (3)
(3)
Copper nuclei, with the exception of the stable isotopes Cu63 and Cu65, decay with positron (e+) and neutrino (ν) emission in Ni nuclei according to the scheme
![]() (4)
(4)
A process alternative to (4), electron capture, in abbreviated form indicated as EC, consists in the nuclear capture of an orbital electron which gives rise to the process
![]() (5)
(5)
As a consequence, in this case, the reaction (4) must be replaced by
![]() (6)
(6)
with emission of an antineutrino.
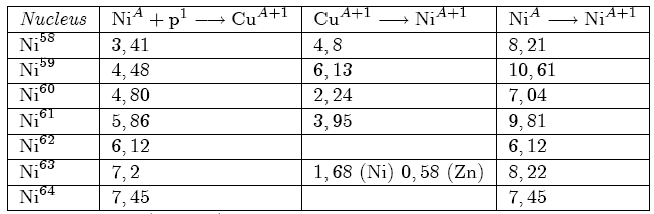 Table 2: Energy (in MeV) released by Ni->Cu and Cu->Ni transformations for different Ni isotopes.
Table 2: Energy (in MeV) released by Ni->Cu and Cu->Ni transformations for different Ni isotopes.
The two decay processes (positron emission and EC) are alternative: their relative frequencies for the various copper isotopes are generally unknown with the only exception of Cu64 for which EC decay (6) is about twice as frequent as positron decay [4].
The capture rate of protons by Nickel nuclei cannot depend on the mass values of different isotopes: in fact they possess the same nuclear charge and the same distribution of electrons in the various atomic shells. In practice, starting from Ni58 which is the more abundant isotope, we can obtain as described in the two above processes, Copper formation and its successive decay in Nickel, producing Ni59, Ni60, Ni61 and Ni62. Because Cu63, which can be formed starting by Ni62 is stable and does not decay in Ni63, the chain stops at Ni62. In Table 2, for every Nickel isotope, we report, expressed in MeV, the energies obtained from the process Ni(A)+p -> Cu(A+1) (column 2), those obtained from the process Cu(A+1) -> Ni(A+1) (column 3) and their total for the complete transformation Ni(A) -> Ni(A+1) (column 4). Data reported in columns 2 and 3, are obtained as differences between the mass values of the initial and final state: that reported in column 3 contain also the neutrino (or antineutrino) energy, particles which interact weakly with the matter and does not hand their energy locally.
On the other hand we have to consider the energy equivalent of the electron rest mass due to the positron annihilation. Cu64 also decays in Zn64 with negative electron emission; the energies relative to both decays are reported in Table 2 (third column); the value carried in column four takes into account the relative frequencies of both Cu64 decay modes. The two isotopes Ni59 and Ni63 are unstable, but because their long lifetime (8×10ˆ4 years and 92 years respectively for Ni59 and Ni63) can be considered as stable in the times of our experiments.
Ni64, coming from the decay of Cu64, decays with electron emission, releasing 2,14 MeV: such a value must be added to 8,22 MeV reported in Table 2 (line 6, column 4).The two isotopes Ni59 and Ni63 are unstable, but because of their long lifetime (8×104 years and 92 years respectively for Ni59 and Ni63) can be considered as stable in the times of our experiments.
For every nucleus in the mass range 58 – 64 amu, we have built Table 3 which contains:
- the mass value expressed in amu (column 1)
- the total energy obtainable from all transformations (column 2)
- the percentage in natural composition (column 3)
- the product of columns 2 and 3
The sum of the energy releases in the last column gives ≈ 35 MeV, which represents the mean energy value obtainable for every Ni nucleus (in the hypothesis that all nuclei give rise to the whole sequence of events).
Such a figure must be compared with E ≈ 200 MeV for every U235 fission in a nuclear reactor [5] and ≈ 18 MeV for every reaction between deuterium and tritium in not still existing fusion reactor.
For the same number of nuclei, the ratio between Ni and U masses is 0,25 and the ratio between the energies that can be obtained is ≈ 0,2. Taking into account the world reserves of these elements, their extraction costs and the great investments needed for the building and maintenance of a nuclear reactor, the nuclear processes (based on Nickel) appear on the economical point very interesting.
During experimental tests, continuous controls on the radioactivity levels in close proximity to the apparatus suitably lead shielded, were performed by using a gamma ray detector [6] and three passive neutron bubble detectors BTbubble [7], one of which for thermal neutrons: no radiation was observed at levels greater than natural radiation background. No radioactivity has been found also in the Nickel residual from the process.
The 10th of march 2009, during the run whose data are reported in Table 1, line five, measurements were performed, around the running Energy Amplifier, by the Bologna University Health Physics Unit which verified that emissions around the Energy Amplifier are not significantly different from the natural background. The water drawn from the Energy Amplifier has resulted to have the same concentration of natural radioisotopes of the tap water: therefore there is no difference between the tap water and the water from the Energy Amplifier.
Two different samples of material used in the experiments labelled in table 1as method A (288 kWh produced) and method B (4774 kWh produced) were analysed at Padova University SIMS. In the long period sample, the mass analysis showed the presence of three peaks in the mass region 63-65 a.m.u. which correspond respectively to Cu63, elements (Ni64 and Zn65) deriving from Cu64 decay and Cu65.
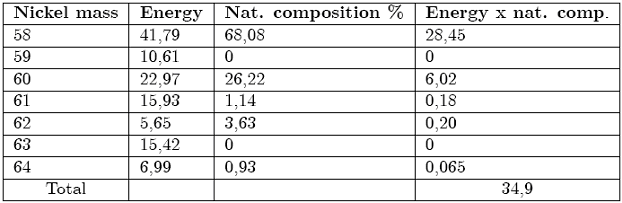 Table 3: Energy obtained by every Ni isotope due to all successive transformations
Table 3: Energy obtained by every Ni isotope due to all successive transformations
These allowed us the determination of the ratio Cu63/Cu65=1,6 different from the value (2,24) relative to the copper isotopic natural composition. The Zn64 derives from the β‾ Cu64 decay: as it.s shown in Table 3, formation of Cu64 requires the existence of Ni63 which, absent in natural Ni composition, must have been in precedence produced starting by more light nickel isotopes. More details on this analysis will be given in a successive paper [8].
Sergio Focardi
Andrea Rossi

Dear Pierre Clauzon:
Tyhank you.
See you end Oct.
Warm Regards,
A.R.
Dear Andrea,
I hope that the building of your 1MW set up is going well and that the final thermal tests will be definitively conclusive… Waiting for this issue with many other friends, I send you this quotation taken from an anonymous writer of a nuclear shielding book: (tranlated from french)
“Doing something causes the open opposition of those who should have done it, those who wanted to do it … but in another way, and of universal criticizers.
Fortunately, there are those who do not say anything but approve and help effectively, sometimes.”
Warm regards
Pierre
Dear Prof. Clauzon:
I will publish the theory behind my technology and the data you are asking about after the presentation of the 1 MW plant of Athens, which is the sole focus of my work now. I want to avoid theoretical confusions and polemics before an undisputable plant is in operation.
We are making a strong work on the isotopes with our USA Partner in this period, and the theoretical puzzle is coming up very well.
Thank you very much for your kind attention, please say hello to my friend Dufour.
Warm regards,
Andrea Rossi
Dear Andrea,
All my wishes for your 1MW set-up in Athens from your always supporters Jean de Lagarde and Pierre Clauzon… Could you tell us if you have now the results about your lengthy experiment and concerning isotopic analysis of Ni and may-be Cu from Padua University, as you wrote to us at mid-2010?
Thanks in advance for your answer and best regards
Pierre
Thank you for your kindly answer, Ing. Rossi. Then, now we can only wait and.. hope ! But, tray please to go at website http://www.leap2020.eu/ (Laboratoir Européen Anticipation Politique), and read their anticipation review : they are very serious people, and their previews for october 2011 are not very good, indeed. Best regards and winning job.
Dear Mr Rolando:
We will not make any further press release or press conference or demo until our 1 MW plant of Athens will be in operation. I think we gave all the possible information so far. At the end of October the diffusion in the market will start.
The fact that we will start in october is due to the time we need to be ready. Now we are not ready for the market and we already geve all the information we can give that is not confidential.
I want to thank you for your important attention; just please consider that we are making the maximum effort we can.
Warm Regards,
A.R.
Dear Andrea, I understand what it means hard-work, and I am sure that your job is very very difficult to do. But I hope that you have the time to read newspapers, about the situation on the world today. It woud be very important to talk about Rossi-Focardi engine NOW ! To explain the hope, for the life of everybody, linked to it. Can you tell something more about your work to the medias ? Wish you to communicate, to peoples on the world, that maybe it exist an alternative, to uranium and petrol energy ? May you organise some press conferences on this subjet ? NOW is the right time to do this ! NOW the attention of the world will be very hight !!! Moreover, Prof. Focardi and you ad all your team, have did a lot of work yet, and you have a lot of results to present. Think about this, please. In october, the situation woud be worst then today. Ciao.
Dear Rolando:
We are working 16 hours per day to arrive in October with our first plant in Athens. You know I am working exclusively with my money, we want not funds from anywhere to avoid discussions. Believe me, we are as fast as possible.
Andrea Rossi
Dear Ing. Rossi,
I think that it is time to show-up your engine. The world cannot wait more. Japan’s situation, about traditionals nuclears plants’ crisis, explains to everybody that is time to change. Can you to do something now, for growing-up the information about your system ? Why not to exhibit NOW the single element of your engine (the 20 kw element)that you own yet ? Come on Andrea, be brave ! We all need it ! Best regards and good work.
Dear Mr Zhengang:
I am convinced that the necessity of new energy sources will allow integrations beyond contrasts. Thank you for your kind attention,
A.R.