Introduction
It is well known that in chemical reactions, and more specifically in processes used to obtain energy, as for example oil, gas and carbon combustion, only some electronVolts (eV) can be obtained for every couple of atoms involved. This depends on the fact that binding energies of external atomic electrons are in the eV range.
On the other hand, in nuclear transformations, the energy quantities that can be absorbed or released are of the order of mega-electronVolts (MeV) for every couple of nuclei involved in the process. As a consequence, for every given amount of energy obtained, the mass to be transformed by a nuclear process is about a millionth of that necessary for a combustion.
It is a general rule, valid for all stable compounds, that the mass for a compound is lower than the total mass of all constituents. In such conditions, the mass-energy conservation principle guarantees stability against the spontaneous disintegration into the components. As a consequence, for the nuclei, the mass
of every stable nucleus turns out to be lower than the sum of the masses of all its components (protons and neutrons).
If we denote by mp and mn the mass values of free protons and neutrons, and by np and nn the numbers of protons and neutrons belonging to a given (stable) nucleus N, the nuclear stability is insured by the always positive difference
![]() (1)
(1)
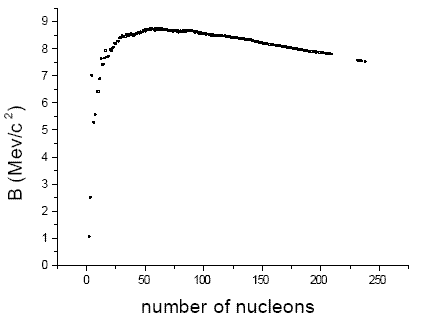 Figure 1: Binding Energy versus number of nucleons
Figure 1: Binding Energy versus number of nucleons
where mN represents the nucleus mass.
An important parameter, whose value is directly connected to the nuclear stability, is the binding energy for a nucleon B [1], defined as the ratio between Δ and the mass number, that is the total nucleon number np+ nn:
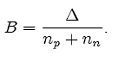 (2)
(2)
Fig.1 shows, for all stable nuclei, the binding energy B (expressed in Mev/cˆ2) versus the total number of nucleons (protons and neutrons) [2].
As is evident from the definition of B, nuclear stability is characterized by large values of the binding energy for nucleon. Nuclei having a mass number around 60 (as Fe, Co and Ni) are characterized as particularly stable.
Fig.1 shows clearly the two existing possibilities in order to obtain energy from nuclear transformations: they consist in producing more stable nuclei starting from low mass or from high mass nuclei. Such two processes are respectively referred to as fusion and fission.
Fusion processes occur naturally in the stars, where helium and other elements are produced, starting from hydrogen. Other similar phenomena, which lead to the production of heavier elements, occur in hydrogen rich stellar atmospheres, after supernovae collapse.
Artificial fission processes are obtained in nuclear reactors by means of neutron interactions with Uranium or Thorium which induce nuclear breaking and neutrons release. There exist no natural fission processes, with the only exception of a flooded Uranium mine in Gabon [3] which reproduced, about two millions years ago, physical conditions similar to the ones occurring in a nuclear reactor.
Sergio Focardi
Andrea Rossi

Dear Robert Wiessner,
Of course, it’s a joke.
I just wanted to make a hyperbolic comment, with irony.
Warmest Regards,
A.R.
Dear Dr. Rossi! I am a regular reader of your blog, but usually I never write comments. Your statements about hornets is in reality related to an urban legend and very old student joke about bumblebees. The German Wikipedia has even a section dedicated to this „bumblebee don’t fly joke“ in their Bublebee article.
In the 1930ies, students of professor Ludwig Prandtl at the university of Göttingen (Germany) invented this joke while sitting in a bar, but could not prevent that it spread over and over and got finally in newspaper articles. Of course no scientist ever was or is now convinced that bublebees can’t fly. Nobody proved that bumblebees can’t fly. The students used only a simple and faulty mathematical model that wasn’t appropriate for describing the flight of a bumblebee.
This myth was also the base of a book with the title „Bumblebees Can’t Fly“ by Barry Siskind.
To calm everybody: someone wrote also a sort of answer book: Robert Cormier: ‚‘The Bumblebee Flies Anyway‘‘.This joke There are many other legends and jokes like this around: for instance the assertion, that chicken would not eat enough calcium carbonate to produce eggs. Believer were French chemist Louis-Nicolas Vauquelin (1763 – 1829) and French chemist and author Corentin Louis Kervran (1901 – 1983), the inventor of an own „biological transmutation“ hypothesis.
Con saluti sinceri, Robert Wiessner
Dear Mr Marco Gentle:
Please contact us in November for commercial issues.
Warm regards,
A.R.
Dear Mr Rossi
Congratulations on your world-changing invention. I have been following the cold-fusion (lenr-canr) field almost since its inception. I am also aware of all the resistance it has been met with. The new media has provided an alternative communication system which will allow the new energy revolution to be met with success. I would like import this technology into South Africa as soon as this could be arranged. Africa has been excluded from so many exciting fields in the past, I hope you consider us too. Energy is something we desperately need out here.
Kind regards
Marco Gentle
Dear Mr Robin van Spaandonk:
Thank you for the suggestion,
I will study on it,
Warm regards,
A.R.
Dear Sir,
If you are interested in alternative methods of converting nuclear power into electricity, then you might consider getting one of your staff to try a little experiment with an E-cat along the lines of Alfred Hubbard’s generator (See http://www.rexresearch.com/hubbard/hubbard.htm ), though this may require using only materials that don’t shield magnetic fields.
Regards,
Robin van Spaandonk
Dear Mr Robin van Spaandonk:
We are in contact in particular with a US turbine manufacturer who is designing a turbine fit for our E-Cats.
Warm regards,
A.R.
Dear Sir,
You wrote:
“Dear Mr Robin van Spaandonk:
Not yet, we are working on it.
Warm regards,
A.R.”
Perhaps if you contact a turbine manufacturer, they would agree to do the work for you, if it meant they might look forward to a possible future partnership? (Since it looks like you will be taking the energy generation market by storm, and such a partnership would be very much in their interest. 😉
Regards,
Robin van Spaandonk
Dear Mr Robin van Spaandonk:
Not yet, we are working on it.
Warm regards,
A.R.
Dear Sir,
I was wondering if you have tried using the steam produced by an E-Cat to drive a turbine and produce electricity, just experimentally? If so, then is there anything you could tell us about the results?
Regards,
Robin van Spaandonk
Dear Mr Ake Ostlund:
1- when the powder has to be changed, as you correctly suggest, the hydrogen tube has to be disconnected
2-I neve insufflated Nytrogen. That info was wrong. If there is a black out, the E-Cat automatically stops, for lack of current: it is intrinsecally safe
Warm Regards,
A.R.
Dear Mr. Rossi,
I am, just like many others, very fascinated by your invention. Personally, because of my profession, I’m particularly interested in safety issues, and out of what I’ve read, I’ve a few considerations concerning issues that might be of relevance in the case of the E-cat – in particular applicable on the household version:
1. In Sweden there are, as in the whole EU I think, rules for how to handle gas tubes – especially those which contains explosive material. As I’ve understood, the consumption of hydrogen is very small, and this machine has to be served twice a year with new Ni-powder. Is it possible to remove the hydrogen tube from the machine and restore the pressure only when serving it? It’d be an advantage if one can have the hydrogen tube removed from the household during the periods when service isn’t required.
2. I’ve read that you once had to stop the machine by insufflating nitrogen and that the E-cat has to be steered or controlled by electricity. What would happen if there’s a break in the supply of electricity – would it then be necessary to stop the machine, and if so, how? Is there a need for an alternative supply of electricity to step in, in form of a (rechargeable) backup battery, in such situations? And would it be a safety step to replace the hydrogen tube with a nitrogen ditto in order to stop the process by an automatic valve which opens up when needed – for ex if the machine becomes overheated?
Best wishes,
Åke Östlund
Dear Sir:
You can email to
info@leonardocorp1996.com
Warm regards,
Andrea Rossi
Dear M. Rossi,
I am a reporter preparing an article about clean energy technologies for a US publication. Our deadline is near and I have just now discovered your work. Please let me know how to communicate with your patent agent or Leonardo in a more private way so that I may ask one or two questions for clarification before we publish our article. I guarantee that I will not share your contact information with anyone.
Thank you,
Ben
Thanks for an interesting article. After looking through different websites I finally found something worth reading.
I hope you will keep updating your content constantly as you have one dedicated reader here.
[…] Introduction […]
[…] Introduction […]
Yes, all the publications on the Journal Of Nuclear Physics are Peer-Reviewed by at least one University professor of Physics expert of the matter of the paper.
We also make the articles reviewed by an attorney, to check the respect of the law.
Warm Regards,
Andrea Rossi
Dear Mr.Rossi.
Thanks a lot for your comprehensive answer.
I’d like to ask you another question:
are the publications on your Journal peer-reviewed?
Best regards
John Spencer
Dear Mr. John Spencer:
I take advantage of your question to explain some principles connected with the methodology of my work.
I imagine you read the Focardi-Rossi Paper, published in this Journal. In that paper there are theoretical explications with the relative calculations. More calculations will be published, after the tests we are doing in this period with the reactor we have in operation in the USA. We are collecting data which will allow us to strengthen the theoretical bases of our work.
But I want to point out that my approach to this job has not been theoretic, and still it is not.
I am working on the attempt to get energy from nuclear weak reactions since 23 years and during these years I studied hundreds of articles and of theoretical calculations which disputed strongly one against the other about the best theoretical approach, giving reciprocal demonsrtration that this couldn’t work for a lot of theoretical reasons, that couldn’t work for an anti-lot of reasons and so on. All, of course, waiting for financing to back the real path to the Holy Graal of the cold fusion. It was clear to me that this policy could bring to nothing. I use to call it the ‘logosturbation syndrome’.
I want to tell you a story. In past some experts said that the hornet has wings too small to fly, and a frequency of wings waving too low to flight, related to the area of the wings and the weight of the hornet. So, based on these calculations, the Hornet CAN NOT FLY. Tell him.
I started from the calculation of the effect of pressure. About the particular way to use the pressure of Hydrogen I have to save confidentiality. I was convinced that with an appropriate use of pressure was possible to have nuclear reactions, along the way described in the calculations you can find in the Focardi Rossi paper. After this I tried, and made thousands of models, working tens of thousands of hours, financing all with my money because I believed in what I was doing, until I reached a result. The result, eventually developed in the patented apparatus, is that we get more energy at the output than the amount of energy we consume at the input. The factor of multiplication of energy is much more than 10. Maybe the theoretical interpretation we made is wrong, maybe we will discover that the energy has other motivations, but this is the actual fact: the apparatus is producing more energy than it consumes and is making it without determining radiations ouside the reactor. This is the fact.
This fact has been made fror a couple of years as of today, in a factory, producing heat which has been used and which has allowed to the factory which used it to save 90% in the energy bill. And as, at last, we have understood why the hornet flies, I am sure we also will understand better and more precisely why the patented apparatus works, I mean the theoretic reasons which today we suppose, but are not content with. Anyway, soon we will publish more calculations about the theoretic analysis.
Thank you very much for your attention,
Warm regards,
Andrea Rossi
Dear Mr.Rossi.
Can you please explain the physic models and the calculations of your invention?
Best regards
John Spencer